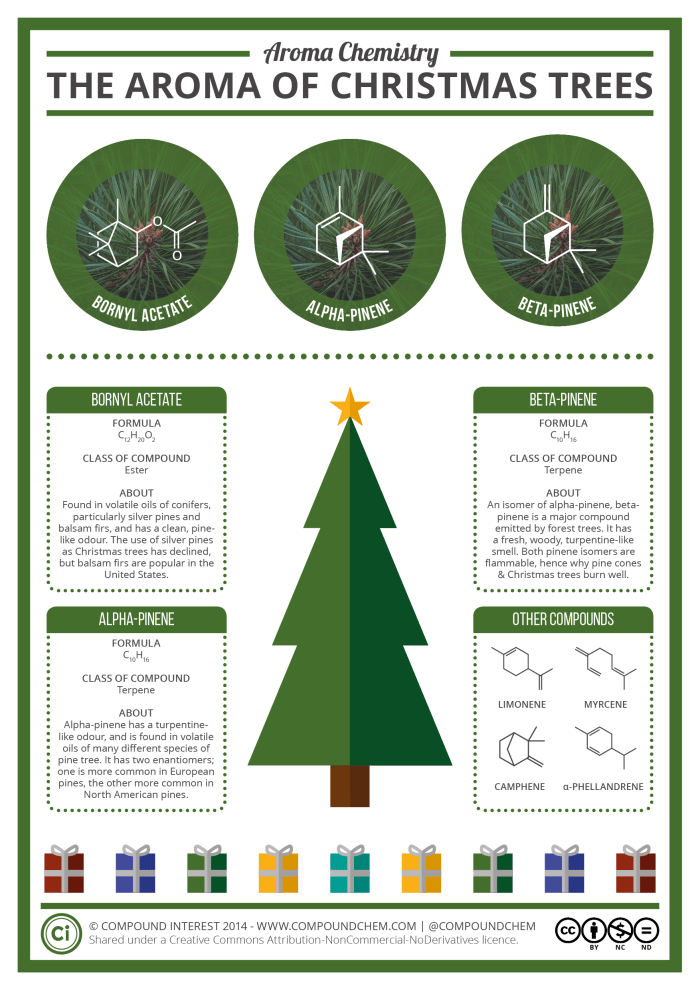
As today marks the past halfway point of this year’s Chemistry Advent Calendar, it seemed like a good point to post a reminder of its existence in case it had slipped under anyone’s radar. You’ll have noticed that other new posts have taken a back seat, as the graphics for the calendar have monopolised the time that I haven’t been spending doing marking for my classes!