
You’ve probably read of the recent death of Stephanie Kwolek, the American chemist who discovered Kevlar. Most people have almost certainly heard of Kevlar, in the context of bullet-proof vests, but fewer are probably aware of the vast range of applications it has, or the chemical reasons behind its strength.
The discovery of Kevlar was almost an accident. Kwolek was working at the chemical company DuPont, and trying to develop a fibre that could be used to make strong but lightweight tyres for cars, when she noticed a mix of some of the polymers she was working with had formed an thin, cloudy solution. This solution was usually thrown away; however she decided to send a sample for tests, and it was found to be much stronger than other comparable fibres such as nylon. Although she was not particularly involved with the subsequent development of applications for Kevlar, these would not have been possible without her discovery.
Kevlar is a polymer – a substance formed from long, chain-like molecules, which are themselves formed from hundreds of smaller molecules, called monomers. The monomers used to form Kevlar are 1,4-phenylene-diamine and terephthaloyl chloride. Kevlar is actually a trade name for the polymer; its chemical name is polyparaphenylene terephthalamide. This is a common occurrence with other polymers as well, with the Teflon found in saucepans and frying pans going by the chemical name polytetrafluoroethene.
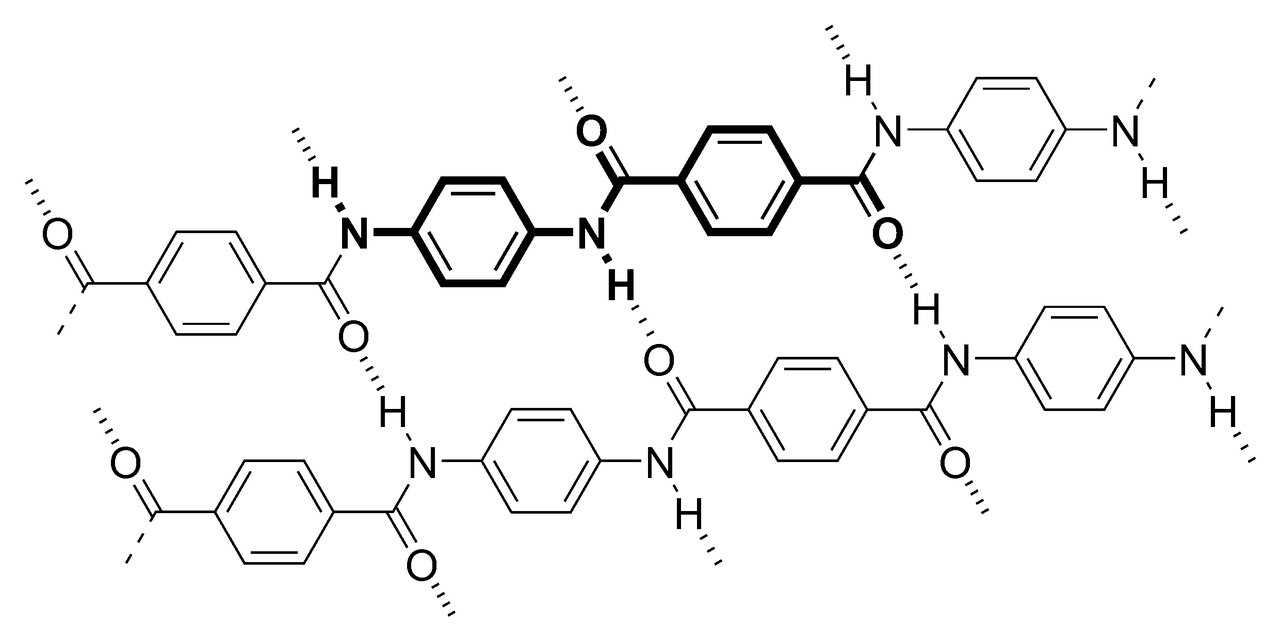
Kevlar’s properties are extremely favourable for its uses. For starters, its density, at 1.44 grams per centimetre cubed is much lower than that of steel, at 7.80 grams per centimetre cubed; this means it is very lightweight by comparison. It’s also five times stronger than steel, on an equal weight basis. Its strength comes from strong intermolecular forces between adjacent chains of the Kevlar polymer. This strong intermolecular force is called ‘hydrogen bonding’, the electromagnetic attraction between some hydrogen atoms in one strand and the oxygen atoms in another. Additional strength is derived from ‘stacking interactions’ between the hexagonal aromatic rings in adjacent chains. Unlike many other plastics, it decomposes at the relatively high temperature of between 427-482˚C.
One of Kevlar’s only weaknesses is its compressive strength. It’s tensile strength (the stress that a material is able to withstand whilst it is being stretched) is very high, but its compressive strength (the stress it can withstand whilst being crushed) is about 1/10 of this. As such, it isn’t much use for applications that demand a high compressive strength, such as building materials.
Most are probably aware of Kevlar’s application in bullet-proof vests and body armour, but it also has a myriad of other uses. It’s used for panels and wings in fighter planes, notably the Eurofighter, and also in the petrol tanks for Formula 1 cars, to prevent rupturing of the tank on crashing. It’s also used in fire and cut-resistant clothing and gloves for both fire services and general use. Ropes and cables use Kevlar, including those in suspension bridges, as it is stronger than steel and does not corrode. Finally, it also finds uses in sport, where layers are added to ping pong paddles to make them lightweight and improve bounce. It’s also used to string some tennis racquets.
These are just a selection of Kevlar’s many uses – you can check out more on DuPont’s page of Uses & Applications. It’s entirely possible that items you make use of every day contain Kevlar – and for that we have Stephanie Kwolek to thank.
The graphic in this article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. See the site’s content usage guidelines.
References & Further Reading
- Kevlar – Technical Guide – DuPont
- Kevlar – Uses & Applications – DuPont
- Kevlar – Explain That Stuff






3 Comments
Comments are closed.