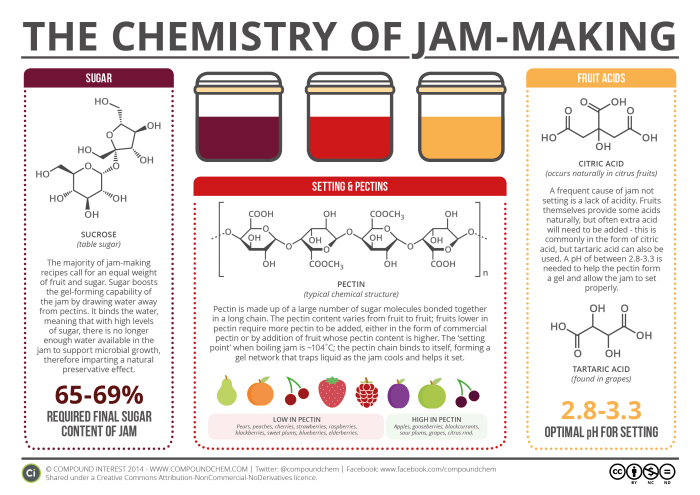
If you’ve ever tried your hand at jam-making, you’ll know that it’s something of a tricky process. A number of factors need to be just right to achieve a perfectly set jam – and chemistry can help explain why. There are three key chemical entities that go into jam-making: sugar, pectin, and acids. Here, we’ll look at each in turn, and how they help jam achieve its eventual consistency.











